
Definition of brick wall efflorescence and building facade
Dissolution of soluble salts in brick or other building materials by water and their deposition on the surface of materials due to water evaporation is called efflorescence.
Efflorescence may appear on the facade of the building at the same time as the building is constructed or sometime after construction is completed.
Primary efflorescence building facade
Materials such as mortar and concrete are used in the construction of the building and its facade. Therefore, when water dries out of the walls and facade of the building, it may cause some efflorescence to form on the surface. This type of efflorescence, which usually occurs in the first few days, is called primary efflorescence.
Secondary efflorescence building facade
Secondary efflorescence includes efflorescence that results from the entry of water into building surfaces and the dissolution of soluble salts and their transfer to the surface of materials.
Types of building efflorescence in terms of chemical nature
There are different types of efflorescence. The most common types of efflorescence include the following:
1- Sulfate efflorescence
2- Carbonate efflorescence
3- Chlorinated efflorescence
4- Nitrate efflorescence
Sulfate efflorescence
After each rain, more or less rainwater seeps into the wall surface, especially in the walls that are most exposed to rain. If there are sulfate salts in the brick, or if the wall is made of gypsum mortar, or it is bonded with gypsum mortar, or water and sulfate-containing mortar are used to make the mortar. The water that penetrates the wall dissolves the sulfate salts and brings them to the surface of the wall. After the water evaporates, the sulfate dissolved in it remains in the form of efflorescence or white on the wall. This continues until all the sulfate salts inside the material appear white on the wall.
In the production of clay bricks, if the iron in the soil is in the form of sulfide (perlite), it is decomposed into iron oxide and SO3 during kiln firing in the kiln. And can cause sulfate efflorescence.
Carbonate efflorescence
Carbonate efflorescence often forms on cementations surfaces such as plasters and mortars. Also, if the brick is made with slurry of lime and cement mortar, when the brick absorbs this slurry or mortar water, calcium hydroxide is transferred from the mortar to the brick. If rain penetrates the wall, the rainwater dissolves calcium hydroxide and carries it to the surface of the wall or facade. After carbon dioxide is absorbed by calcium hydroxide and water evaporates, it remains on the wall in the form of efflorescence or whiteness.
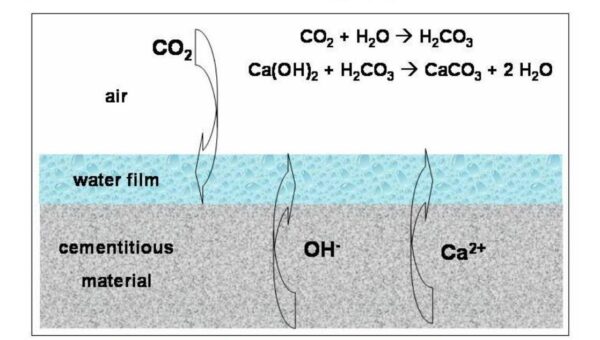

Chlorinated Efflorescence
If diluted salt ink is used to clean the carbonate Efflorescence of the wall facade, then the salt ink combines with the calcium carbonate on the wall surface to form calcium chloride, which must be washed with water.
If the salt ink is thick or not well sprayed, or the facade of the wall is not sprayed and wetted before spraying, the salt ink stays thin at the joints and seams of the wall, sucks the brick and makes calcium chloride with calcium hydroxide mortar. After the wall dries, calcium chloride remains white on the wall.
Also, if stone or brick containing chlorine salts is used in the construction of the wall, or water containing chlorine salts penetrates the wall, chlorine bleach will be poured on it. Calcium chloride is a humidifier. Whenever you do not wash rainwater off the wall, it dampens in humid air and damp spots are found on the wall. After the wall dries, the crystallized calcium chloride remains white on the wall.
Nitrate Efflorescence
When bricks or other building materials, such as mortar, are contaminated with nitrate-containing objects such as decayed mortars before use, their nitrate leaks in moist air and damp spots are found on the wall, which after drying form white nitrate stains. .
The mechanism of Efflorescence in building facade
The following three conditions must be met to create Efflorescence on the facade surface. Efflorescence will not be possible if any of the following factors are removed.
Presence of soluble salts in materials (facade)
Sufficient moisture to convert salts into saline solutions.
Existence of a path for moving salt solutions to the surface of materials and evaporation of water and deposition of salts on the surface of materials (facade)
Factors causing brick Efflorescence in brick facades
The most common cause of Efflorescence on brick surfaces are sodium, potassium, magnesium and calcium sulfates; This is because these compounds are often present in the clay that is the source of brick production or are produced during kiln brick production. These salts can be easily dissolved by rainwater and transferred to the surface of the brick by water to cause whiteness and Efflorescence. These types of Efflorescence often have high solubility in water and their adhesion to the surface is poor.


Another common type of Efflorescence on brick surfaces is carbonating Efflorescence, which is often transferred from mortar to brick.
When Efflorescence appears on the wall, it is often mistaken for a brick. While the salts in the mortar, sand and water used can also cause Efflorescence.
The role of mortar in the formation of brick Efflorescence in building facade
The first and most obvious source that contaminates bricks with Efflorescence is mortar. Mortar is in contact with the brick at least from four sides and sometimes from 5 sides. Mortar is used in a wet state with bricks, in which case it provides a lot of moisture to transfer soluble salts from the mortar to the brick. If the amount of mortar soluble salts is significant, the mortar soluble salts will be transported to the brick in proportion to the amount of moisture transferred.
Reminder:
As mentioned earlier, calcium hydroxide is found in all cementations concrete mortars as a setting additive. After dissolving by rain and transporting to the surface of the brick, it reacts with carbon dioxide in the air to form calcium carbonate, which can appear white on the surface of the brick.
Brick baking temperature:
Brick firing temperature can affect the presence or absence of Efflorescence. When the temperature of a brick kiln rises above 1000 degrees, most of the sulfate salts such as sodium, potassium and magnesium sulfates in the brick are released, while calcium sulfate requires a temperature of at least 1,200 degrees to be released; Therefore, increasing the temperature to 1200 degrees can play an effective role in reducing Efflorescence; Therefore, among the sulfate salts present in the soil, only calcium sulfate will remain in the brick, which has a lower solubility than other sulfate salts.
Improper filling of the space between the bricks when executing the building facade bricks:
If the bonding is not done properly, Efflorescence stains or in other words, bonding mortar will contaminate the entire surface of the facade and this mortar will intensify the Efflorescence.


Carbonate Efflorescence created on brick wall joints
Weather conditions at the time of construction of the building facade brick:
In winter, Efflorescence intensifies due to the cold weather and increased humidity. When a new brick wall is exposed to moisture, the moisture of the soluble elements will rise to the surface. Calcium hydroxide, which is often present in cement and mortar, dissolves more when the temperature decreases and its maximum solubility is at 4 ° C. Therefore, it intensifies carbonate Efflorescence.
Factors affecting Efflorescence on cementations surfaces
White cement is often used in the construction of facades due to its paint ability and the possibility of creating beautiful designs and colors on it; Ordinary cement is often used as a plaster on the surface of walls.
As mentioned earlier, the most important soluble salt in cement mortars and concrete is calcium hydroxide, which reacts with atmospheric carbon dioxide after dissolution and transfer to the surface to produce calcium carbonate. Of course, depending on the quality and type of water used, sand as well as salt materials that may be used as mortar and concrete as a quick setting, it is possible to produce chloride, sulfate and even nitrate Efflorescence on cement surfaces.
Carbonate Efflorescence on white cement surfaces is less common as long as plain (non-colored) white cement, but Efflorescence on colored and patterned surfaces can play an effective role in reducing the beauty of the surface.
Methods for removing Efflorescence if it appears on the building facade
The first step in eliminating Efflorescence is to identify the salt that causes Efflorescence. If water-soluble salt is the cause of Efflorescence, the best way to remove it is to use physical methods.
There are generally two main ways to get rid of Efflorescence.
Physical methods of Remove brick efflorescence
Physical methods of Efflorescence removal include the use of brushes, sandblasting and other methods. The choice of any of these methods will depend on factors such as the type of Efflorescence, the dimensions and material of the surface, the height and accessibility of the surface and the location, and so on.
Chemical methods of Remove brick efflorescence
The use of chemicals is also effective in removing Efflorescence. After using Efflorescence chemical solutions, the wall should be thoroughly rinsed with clean water.
Diluted solutions of acids such as hydrochloric acid, acetic acid (vinegar) and phosphoric acid are often used to dissolve and remove some Efflorescence.
One of the most common solutions used to remove Efflorescence is a solution of hydrochloric acid and water. This solution is used in a ratio of 1 to 12. Identifying the type of salt that causes Efflorescence is very important in removing it, so if the salt that causes Efflorescence can react with hydrochloric acid (such as carbonate salts) and cause other Efflorescence on the facade, the use of other solutions such as phosphoric acid solution can be diluted with water. Be useful.
Cleaning Efflorescence from the wall is not a cure for Efflorescence. This only removes the signs of Efflorescence from the wall and it is possible for it to reappear on the wall unless one of the three conditions causing the Efflorescence is removed. The appearance of Efflorescence on a wall indicates the presence of soluble salts in the wall and the possibility of transferring saline solutions from the wall to the surface. Therefore, water infiltration into the wall and the conversion of solid salts into saline solutions should be avoided. For this reason, after sandblasting or washing the wall with chemical solutions, the wall or facade should be covered with sealing or water-repellent materials.
Ways to control Efflorescence in building facade
Efflorescence control methods can include solutions related to the construction or selection of materials; Or use solutions after finishing work and execution on the facade.
Solutions related to the construction or selection of materials
1- Reduction of alkaline sulfates that make up bricks (including controlling the quality of soil used or baking bricks at higher temperatures)
2- Using materials that are less likely to cause Efflorescence (for example, using silica sand bricks instead of clay bricks)
3- Use bricks whose water absorption percentage is between 10 and 15%.
4- Use drinking water with low hardness and washed sand and minimize the water required when making mortar and cement
Solutions that can be implemented on the facade
Prevent water from penetrating into the wall and facade
A- Use coating or sealing materials and water repellent on the facade of the building to prevent the infiltration of water into the facade
B- Insulation in the lower part of the wall that is connected to the ground
One of the materials that can be used as an impermeable building facade is permeable silicate primers. These primers can be used as sealants for walls and facades of buildings due to having some of the following features.
1- Colorlessness and non-change of color and appearance
2- High penetration power into the surface of various construction materials
3- Ability to remove some salts that cause Efflorescence such as calcium hydroxide in materials
4- Filling the pores and capillaries inside the building materials and preventing water from penetrating into the materials
5 – No water repellent on the surface of the material (this parameter can be very useful for cases where another coating is to be applied on the surface)
6- Mineralogy of these primers and durability and long life
7- Resistant to negative water pressure








